 |
 |
Rhabdomyolysis and Excited Delirium: Different Stages of the Same Syndrome |
CITATION:
Ruttenber AJ, McAnally HB, Wetli CV. Cocaine-associated rhabdomyolysis
and excited delirium: different stages of the same syndrome.
Am J Forensic Med Pathol; June 1999; 20(2), pp 120-127.
CHAS' OPINION of this old (and erroneous) case review report will be reflected in her
2005 Paper: Restraint Asphyxia Research Review
When that paper is posted, the above link will work.
 In the mean time, read the much more recent papers about this subject:
In the mean time, read the much more recent papers about this subject:
Ruttenber, A. James Ph.D., M.D.; McAnally, Heath B. M.S.P.H.; Wetli, Charles V. M.D.
From the Department of Preventive Medicine and Biometrics, University of Colorado School of Medicine, Denver, Colorado (A.J.R., H.B.M.), and the Office of the Chief Medical Examiner, Suffolk County, New York, New York (C.V.W.), U.S.A.
Received February 19, 1998; accepted November 16, 1998.
Address correspondence and reprint requests to A. James Ruttenber, Department of Preventive Medicine and Biometrics, University of Colorado School of Medicine, Campus Box C-245, 4200 East Ninth Avenue, Denver, CO 80262, U.S.A.;
 Email: jim.ruttenber@uchsc.edu
Email: jim.ruttenber@uchsc.edu

Abstract:
Previous case reports indicate that cocaine-associated rhabdomyolysis and excited delirium share many similar features, suggesting that they may be different stages of the same syndrome. We tested this hypothesis by comparing data from 150 cases of cocaine-associated rhabdomyolysis reported in the medical literature with data from an autopsy registry for 58 victims of fatal excited delirium and 125 victims of fatal acute cocaine toxicity. Patients with rhabdomyolysis are similar to victims of fatal excited delirium with regard to age; gender; race; route of cocaine administration; the experiencing of excitement, delirium, and hyperthermia; and the absence of seizures. Compared with victims of fatal acute cocaine toxicity, patients with rhabdomyolysis are different with regard to each of these variables. Compared with victims of fatal acute cocaine toxicity, both victims of rhabdomyolysis and fatal excited delirium are more likely to be black, male, and younger; to have administered cocaine by smoking or injection; and to have experienced excitement, delirium, and hyperthermia; they are also less likely to have had seizures. Because cocaine-associated rhabdomyolysis and excited delirium have similar clinical features and risk factors, occur in similar populations of drug users, and can be explained by the same pathophysiologic processes, we conclude that they are different stages of the same syndrome. It appears that this syndrome is caused by changes in dopamine processing induced by chronic and intense use of cocaine rather than by the acute toxic effects of the drug.
Since the early reports of cocaine-associated rhabdomyolysis in 1987(1,2), numerous descriptions of this syndrome have appeared in the literature. Coincidentally, there have been a number of reports of cocaine-induced excited delirium (3-7), along with evidence of rhabdomyolysis in victims of fatal excited delirium who were hospitalized before death (8). Because these two cocaine-associated conditions exhibit many similar characteristics, including hyperthermia, bizarre, psychotic behavior, and hyperactivity, we proposed that they are different stages of the same pathophysiologic process (9).
In this article, we present further evidence with a comparison between combined analysis of reports in the medical literature for persons with cocaine-associated rhabdomyolysis and data for deaths from excited delirium and acute cocaine toxicity that were collected in an ongoing epidemiologic study of cocaine-related deaths in Dade County, Florida.
CASE REPORT
A 36-year-old white man was found wandering in yards in the Coconut Grove residential area of Miami, Florida. A fire-rescue unit was summoned and, on arrival, the subject became agitated and combative. He was restrained and transported to a local hospital. At admission, his temperature was 39.8�C, pulse was 122, respiration rate was 36 breaths/minute, and blood pressure was 100/55 mm Hg. While hospitalized, he continued to exhibit altered mental status, remained febrile, and had episodic seizures. Within 2 days of admission, he developed rhabdomyolysis, disseminated intravascular coagulation, and hepatic and renal failure. He died 60 hours after admission.
Autopsy revealed a 520-g heart with biventricular hypertrophy and no gross or microscopic evidence of coronary artery disease or myocardial damage. The lungs were hyperinflated and diffusely consolidated. Both kidneys were swollen and enlarged, and microscopic examination revealed proteinaceous deposits in Bowman's space and proximal tubules. The liver was enlarged, and microscopic examination revealed patchy congestion, minimal steatosis, and lymphoid infiltration of portal zones. Toxicologic analysis of an unpreserved blood sample drawn on hospital admission revealed an ethanol concentration of 0.02%, a benzoylecgonine concentration of 0.35 mg/L, and the presence of methylecgonine and ethylecgonine, which are minor cocaine metabolites. Cocaine itself was not detected. Analysis of a sample of white powder taken from the subject at his arrest revealed cocaine with a concentration of 85% (calculated as cocaine base) and the presence of no other drugs.
The subject had a history of cocaine abuse and was known to frequent areas of Coconut Grove where cocaine was sold and used. Members of the subject's family alleged he was poisoned by a drug dealer and a warrant was issued for the arrest of the dealer.
METHODS
An online literature search was performed to find articles on cocaine-associated rhabdomyolysis published in English. We identified 37 papers (including one abstract), 31 of which contained individual case reports for a total of 60 cases (1,2,10-36), and 6 case series comprising 100 cases (37-42). Of the subjects described in the case series, 10 were previously described in case reports and were excluded from the case series data to avoid double counting.
We abstracted demographic and clinical data from these reports and compared them with data for victims of fatal excited delirium and fatal acute cocaine toxicity without excited delirium from a population-based registry of cocaine-related fatalities in Dade County, Florida (9) for 1979 through 1990, the time period during which most of the reported cases of cocaine-associated rhabdomyolysis occurred. The data were compared using ?2 test for categorical variables and Student's t-test for continuous variables. Statistical analyses were performed using SAS v. 6.12 software (43).
RESULTS
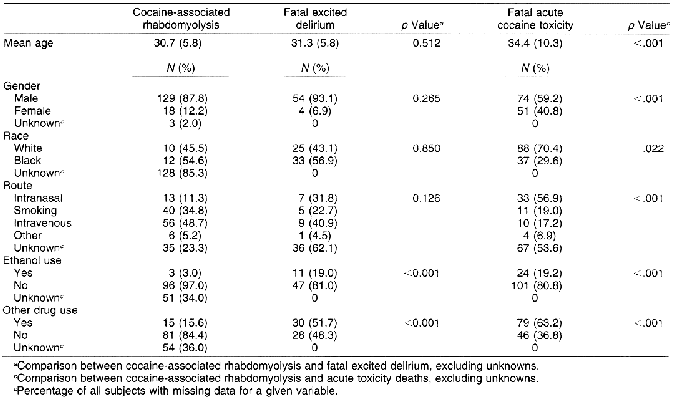
Victims of cocaine-associated rhabdomyolysis and fatal excited delirium were predominantly male and comprised higher percentages of blacks than either victims of fatal acute cocaine toxicity (Table 1), or the Dade County population (in 1990, 20.5% of Dade County residents were black (44). Patients with cocaine-associated rhabdomyolysis were similar to victims of fatal excited delirium with regard to the routes of cocaine administration (Table 1). Compared with victims of fatal acute cocaine toxicity, however, persons with cocaine-associated rhabdomyolysis administered cocaine more frequently by smoking or injection and less frequently by nasal insufflation (Table 1). Ethanol and other drugs were identified less frequently in victims of cocaine-associated rhabdomyolysis than in persons who died of either excited delirium or acute cocaine toxicity.
Hyperthermia was less frequently reported for victims of cocaine-associated rhabdomyolysis than for victims of fatal excited delirium (Table 2). In the medical examiner reports of death and autopsy results, it was the usual practice to note body temperature if it was elevated. If body temperature was within normal limits, it was usually not mentioned and was recorded in the epidemiologic database as unknown (Table 2). In light of this practice, it is likely that most of the fatal acute cocaine toxicity subjects with unknown body temperatures were normothermic at the time of death and that hyperthermia is likely to be less frequent in victims of fatal acute cocaine toxicity than in persons with cocaine-associated rhabdomyolysis.
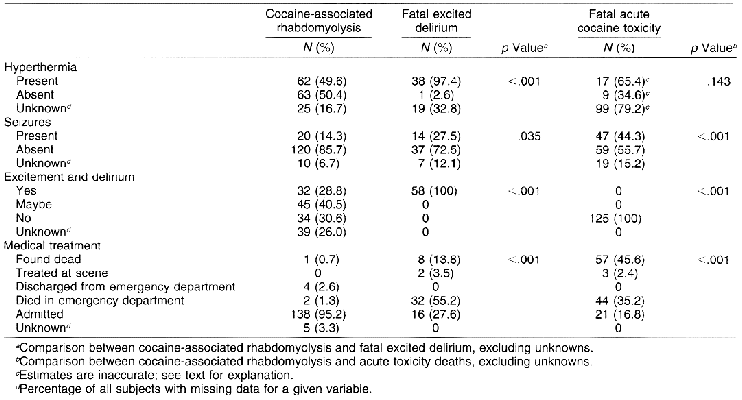
Seizures were less frequently observed among victims of cocaine-associated rhabdomyolysis (14.3%) than among victims of fatal excited delirium (27.5%; Table 2). Both groups experienced seizures less frequently than victims of fatal acute cocaine toxicity (44.3%). Additionally, medical treatment given after the onset of symptoms was more common for victims of cocaine-associated rhabdomyolysis than for victims of excited delirium (Table 2). Finally, the presence of excitement and delirium, by definition, occurred in all victims of excited delirium; a sizable portion of victims of cocaine-associated rhabdomyolysis also exhibited these features.
Strengths and Limitations
To reduce misclassification error, all medical examiner reports were reviewed by a single epidemiologist (A.J.R.) experienced in the diagnosis of the different facets of cocaine toxicity. This was done to ensure consistency between diagnoses of fatal excited delirium and acute fatal cocaine toxicity. Moreover, all data obtained from the Metropolitan Dade County Medical Examiner Department were carefully reviewed and edited.
Data are missing for some of the variables. This points to an area that deserves improvement by clinicians and forensic pathologists who collect and record primary data. We do not think, however, that analyses performed with missing data are subject to bias. In our paper, the most likely affect of missing data is to lower the statistical power of the study to detect significant differences between study groups.
Because subjects came from a variety of locations and some were alive and some dead, it is not possible to assess the percentage of acute toxicity victims who develop rhabdomyolysis, excited delirium, or both. A prospective study in a large metropolitan area emergency department could help clarify the attributable risk for these two outcomes.
DISCUSSION
Common Features
The victims of cocaine-associated rhabdomyolysis are similar to victims of fatal excited delirium with regard to age, gender, and race; both groups are different from victims of acute cocaine toxicity with respect to these demographic variables. In comparison to victims of fatal acute cocaine toxicity, persons with cocaine-associated rhabdomyolysis and victims of fatal excited delirium are less likely to have administered cocaine by nasal insufflation.
In a previous study of victims of fatal excited delirium, Ruttenber et al. (9) linked black race, male gender, and administering cocaine by a route other than nasal insufflation with a pattern of chronic and intense cocaine administration. Many of the articles on victims of cocaine-associated rhabdomyolysis also describe periods of chronic cocaine use or "bingeing" that preceded the onset of rhabdomyolysis (13-15,19,22,24,25,27,28,33,35); other reports identified victims as known abusers of cocaine (12,20,23,36). Taken together, the data suggest that victims of cocaine-associated rhabdomyolysis and fatal excited delirium used cocaine more frequently and intensively than those who died of acute cocaine toxicity.
The connection between cocaine-associated rhabdomyolysis, excited delirium, and chronic cocaine use is also supported by a temporal coincidence between the appearance of these two conditions and the epidemic of crack cocaine use in the United States (45). Crack is associated with chronic "binge" use more often than intranasal administration of cocaine hydrochloride (46,47). In fact, the first report of excited delirium (3) and one of the early descriptions of cocaine-associated rhabdomyolysis (31) came from Miami-the first metropolitan area in the United States to experience an influx of cheap and highly pure cocaine hydrochloride in the early 1980s due to newly established transport routes through the Caribbean (48). Miami was also one of the first cities in which crack was aggressively marketed beginning in 1985.
Victims of cocaine-associated rhabdomyolysis and fatal excited delirium are also similar with regard to having experienced hyperthermia, and different from victims of acute cocaine toxicity, who exhibited this sign less frequently. However, hyperthermia was less frequently reported for victims of cocaine-associated rhabdomyolysis than for victims of excited delirium. Therefore, we speculate either that persons with cocaine-associated rhabdomyolysis survived a period of hyperthermia that was not evident at the onset of rhabdomyolysis and was not reported, or that hyperthermia is not a necessary component for the induction of rhabdomyolysis.
Proposed Mechanisms
If cocaine-associated rhabdomyolysis and excited delirium are components of the same syndrome, then they should share the same initiating factors and have similar pathophysiologic processes. Although several hypotheses have been proposed for cocaine-associated rhabdomyolysis (Table 3), none has considered excited delirium as a component of the disease process.
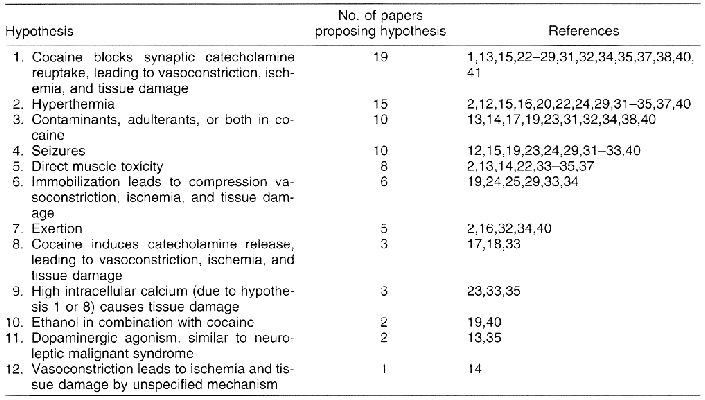
The most commonly proposed mechanism is the blockade of synaptic catecholamine reuptake and induction of adrenergic agonism, resulting in vasoconstriction and ischemia and leading to muscle tissue damage (hypothesis 1). Alternatively, it has been proposed that cocaine induces similar pathophysiology by increasing the release of catecholamines (hypothesis 8). Some have suggested that these processes actually induce tissue damage by increasing concentrations of intracellular calcium (hypothesis 9), or by vasoconstriction of unspecified etiology (hypothesis 12). Although each of these mechanisms may help explain the induction of rhabdomyolysis, none explains the onset of excited delirium. Furthermore, these mechanisms would not be expected to be differentially associated with the aforementioned risk factors of gender, race, hyperthermia, and administration of cocaine by smoking or injection.
Although compression vasoconstriction has been proposed as a mechanism (hypothesis 6), only a few victims of cocaine-associated rhabdomyolysis had histories of coma or prolonged unconsciousness that could account for this effect. Seizures (hypothesis 4) likewise are not probable, as our data demonstrate that they occurred far less frequently in victims of cocaine-associated rhabdomyolysis and victims of excited delirium than in victims of fatal acute cocaine toxicity.
Ethanol in combination with cocaine (hypothesis 10) is also an improbable explanation, because ethanol was less commonly reported for victims of cocaine-associated rhabdomyolysis or detected in victims of fatal excited delirium than it was detected in victims of fatal acute cocaine toxicity. Furthermore, a recent emergency department report has shown that the incidence of rhabdomyolysis and mean blood urea nitrogen concentration were lower in patients consuming both cocaine and ethanol than in patients who used cocaine exclusively (49).
Contaminants or adulterants in street preparations of cocaine (hypothesis 3) are not likely either, because complete toxicologic analyses of autopsy blood samples from victims of excited delirium identified no drugs or chemicals other than cocaine that could have induced excitement, delirium, or rhabdomyolysis (9). Moreover, drugs other than cocaine were detected less frequently in autopsy blood samples from victims of excited delirium than from persons who died of acute cocaine toxicity.
A direct toxic effect of cocaine upon muscle tissue (hypothesis 5) has been proposed based on evidence from animal models. Cocaine has been shown to induce creatine kinase leakage from muscle tissue, although the mechanism for this damage has not yet been elucidated (50,51). It is not clear, however, how direct toxicity can be related to the aforementioned risk factors for cocaine-associated rhabdomyolysis-unless muscle toxicity is correlated with chronic and intense cocaine use, as mentioned previously. Moreover, this mechanism would not induce excitement or delirium.
A period of exertion (hypothesis 7) preceded death in all of the victims of excited delirium and was noted for several cocaine-associated rhabdomyolysis subjects in the published studies we reviewed. Exertion is known to induce rhabdomyolysis, and it is reasonable to assume that males and young persons would be more likely than others to exhibit this sign. It is not clear why blacks would be at increased risk for exertion compared with whites, unless it is because they are at higher risk for chronic and intense cocaine use (9), as discussed later. In the reports of cocaine-associated rhabdomyolysis we reviewed, there was no information on why victims experienced periods of exertion.
A number of the persons in reported cases of cocaine-associated rhabdomyolysis exhibited normal or lethargic states preceding the onset of rhabdomyolysis. It is not clear whether these states were preceded by unreported or unrecorded periods of exertion or whether exertion is not a requisite for cocaine-associated rhabdomyolysis.
One of the most commonly proposed hypotheses is that hyperthermia induces rhabdomyolysis (hypothesis 2). Our data also identify hyperthermia as an important feature of excited delirium, and we have discussed previously how this clinical sign may provide important clues to the mechanism for excited delirium (9). Hyperthermia is a prominent feature of the neuroleptic malignant syndrome, and Daras et al. (35) pointed out the similarities between this syndrome and cocaine-associated rhabdomyolysis (hypothesis 11). Both neuroleptic malignant syndrome (52,53) and excited delirium (9) occur more frequently in summer months than at other times of the year, suggesting that ambient temperature and humidity play roles in the evolution of hyperthermia. Although no data on season of occurrence or ambient temperature and humidity were reported for victims of cocaine-associated rhabdomyolysis, it is interesting to note that of the 150 subjects in the 36 papers reviewed, 47% occurred in either Miami or Atlanta, 26% in New York City, and 7% in other southeastern states-areas where periods of high temperature and humidity are common.
Both hyperthermia and hyperactivity appear to play important roles in the evolution of cocaine-associated rhabdomyolysis and excited delirium. The high proportion of victims of cocaine-associated rhabdomyolysis admitted to hospitals and surviving compared with the low rate of hospitalization for deaths from excited delirium suggests that prompt medical treatment may reduce both the risk for death from hyperthermia as well as the risk for rhabdomyolysis. Because hyperthermia was noted in about 50% of patients with rhabdomyolysis, we surmise that it is a sufficient but unnecessary cause. We caution, however, that hyperthermia could have been substantially underreported in this group. It is possible that exertion without hyperthermia accounts for a portion of the normothermic patients.
Chronic Cocaine Use and Dopamine Processing
In neuroleptic malignant syndrome, the elevated risk for hyperthermia is thought to result from changes in dopamine processing precipitated by the chronic administration of neuroleptic drugs, not from acutely elevated or lowered drug concentrations. The hyperthermia of neuroleptic malignant syndrome is associated with psychomotor agitation, and both have been related to localized increases in dopamine concentrations. Both of these effects are thought to be initiated by the chronic blockade of dopamine in the different neural pathways involved in both thermoregulation and neuromuscular homeostasis (54).
Although studies of the adaptive effects of chronic cocaine administration have been inconsistent (perhaps owing to different animal models and differences in the regions of the brain that were studied), most have shown some change in both the number and function of dopamine receptors, dopamine transporters, or both (55-60). The density and sensitivity of D1 and D2 receptor subtypes are altered by drugs that increase intrasynaptic dopamine (61). In humans, chronic cocaine use decreases the density of D1 receptors in the striatal reward centers, but does not affect the number of D2 receptors (62). Among victims of fatal excited delirium, reduction in the number of D2 receptors was noted within temperature regulatory centers of the hypothalamus. Because these receptors decrease core body temperature, such a reduction could lead to hyperthermia (62).
Aberrant processing of dopamine can also explain the onset of the other features of cocaine-associated rhabdomyolysis and excited delirium. Compared with drug-free controls, cocaine recognition sites on the striatal dopamine transporter are increased in cocaine users without excited delirium (63). A similar increase was not seen in victims of excited delirium (64), suggesting that they may have problems clearing synaptic dopamine, leading to agitation, exertion, and delirium.
There is additional evidence that supports the hypothesis that chronic cocaine use can produce long-lasting changes in dopamine processing that results in both excited delirium and rhabdomyolysis: creatine kinase levels have been shown to be elevated both in asymptomatic current cocaine users (65) and in a group of former chronic cocaine users who have abstained from cocaine use for an average period of 3 months (66), and cocaine-induced paranoid psychosis (a condition similar to excited delirium) persists long after cessation of chronic cocaine use (58).
CONCLUSIONS
Our evidence supports the hypothesis that cocaine-associated rhabdomyolysis and excited delirium are features of the same syndrome, and that chronic cocaine use rather than acute toxicity is responsible for this syndrome. This connection has been difficult to recognize because excited delirium is usually noticed by medical examiners due to its unique combination of bizarre behavior, hyperthermia, and cardiac arrhythmia causing death before rhabdomyolysis can develop. Those who survive this phase are at risk for rhabdomyolysis, which is diagnosed by emergency medical personnel or by urologists some time after excitement, delirium, and hyperthermia have resolved.
By recognizing excited delirium and cocaine-associated rhabdomyolysis as facets of the same syndrome, we can learn more about each condition from the other. The autopsy toxicology data for excited delirium deaths have helped to clarify that this syndrome is not precipitated by acutely high concentrations of cocaine or other drugs and that it appears to be caused by changes in dopamine receptors and transporters-data that are not routinely available for victims of cocaine-associated rhabdomyolysis. Conversely, interview data regarding victims of cocaine-associated rhabdomyolysis have helped verify the hypothesized risk for chronic cocaine use. Perhaps even more can be learned from interviewing those who have experienced the syndrome and lived.
A number of questions about excited delirium and cocaine-associated rhabdomyolysis remain to be answered. Foremost is why only certain chronic cocaine users are at risk for this syndrome. Because of the similarities in presentation and pathophysiology, it appears that the answer to this question is closely related to why only some medicated schizophrenics are at risk for neuroleptic malignant syndrome, and why some unmedicated schizophrenics appear to be predisposed to excitement and delirium.
Acknowledgment: Partial support for this research was provided by grant DA 06227 from the National Institute on Drug Abuse.
REFERENCES
1. Schwartz JG, McAfee RD. Cocaine and rhabdomyolysis. J Fam Pract 1987;24:209.
2. Merigian KS, Roberts J. Cocaine intoxication: hyperpyrexia, rhabdomyolysis and acute renal failure. Clin Toxicol 1987;25:135-48.
3. Wetli CV, Fishbain DA. Cocaine-induced psychosis and sudden death in recreational cocaine users. J Forensic Sci 1985;30:873-80.
4. Loghmanee F, Tobak M. Fatal malignant hyperthermia associated with recreational cocaine and ethanol abuse. Am J Med Pathol 1986;7:246-8.
5. Jaffey PB, Haque AK, el-Zaatari J, Pasarell L, McGinnis MR. Disseminated conidobolus infection with endocarditis in a cocaine abuser. Arch Pathol Lab Med 1990;114:1276-8.
6. O'Halloran RL, Lewman LV. Restraint asphyxiation in excited delirium. Am J Forensic Med Pathol 1993;14:289-95.
7. Mirchandani HG, Rorke LB, Sekula-Perlman A, Hood IC. Cocaine-induced agitated delirium, forceful struggle, and minor head injury. Am J Forensic Med Pathol 1994;15:95-9.
8. Mayuri P, Wetli CV. Sudden death from cocaine-induced excited delirium: an analysis of 45 cases. Am J Clin Pathol 1995;104:329.
9. Ruttenber A, Lawler-Heavner J, Yin M, Wetli C, Hearn W, Mash D. Fatal excited delirium following cocaine use: epidemiologic findings provide new evidence for mechanisms of cocaine toxicity. J Forensic Sci 1997;42:25-31.
10. Rhabdomyolysis (clinical conference). N Y State J Med 1988;88:582-8.
11. Barrido D, Joseph A, Rao T, Friedman E. Renal disease associated with acute and chronic "crack" abuse (abstract). Kidney Int 1988;33:181.
12. Campbell B. Cocaine abuse with hyperthermia, seizures and fatal complications. Med J Aust 1988;149:387-9.
13. Herzlich BC, Arsura EL, Pagala MP, Grob D. Rhabdomyolysis related to cocaine abuse. Ann Intern Med 1988;109:335-6.
14. Krohn KD, Slowman-Kovacs S, Leapman SB. Cocaine and rhabdomyolysis. Ann Intern Med 1988;108:639-40.
15. Lombard J, Wong B, Young JH. Acute renal failure due to rhabdomyolysis associated with cocaine toxicity. West J Med 1988;148:466-7.
16. Menashe PI, Gottlieb JE. Hyperthermia, rhabdomyolysis, and myoglobinuric renal failure after recreational use of cocaine. South Med J 1988;81:379-81.
17. Reinhart W, Stricker H. Rhabdomyolysis after intravenous cocaine. Am J Med 1988;85:579.
18. Skluth H. Rhabdomyolysis associated with cocaine intoxication. Drug Intel Clin Pharm 1988;22:778-80.
19. Anand V, Siami G, Stone WJ. Cocaine-associated rhabdomyolysis and acute renal failure. South Med J 1989;82:67-9.
20. Bauwens JE, Boggs JM, Hartwell PS. Fatal hyperthermia associated with cocaine use. West J Med 1989;150:210-2.
21. Guerin J, Lustman C, Barbotin-Larrieu F. Cocaine-associated acute myoglobinuric renal failure. Am J Med 1989;87:248-9.
22. Jandreski MA, Bermes EW, Leischner R, Kahn SE. Rhabdomyolysis in a case of free-base cocaine ("crack") overdose. Clin Chem 1989;35:1547-9.
23. Parks JM, Reed G, Knochel JP. Case report: cocaine-associated rhabdomyolysis. Am J Med Sci 1989;29:334-6.
24. Pogue VA, Nurse HM. Cocaine-associated acute myoglobinuric renal failure. Am J Med 1989;86:183-6.
25. Singhal P, Horowitz B, Quinones MC, Sommer M, Faulkner M, Grosser M. Acute renal failure following cocaine abuse. Nephron 1989;52:76-8.
26. Steingrub JS, Sweet S, Teres D. Crack-induced rhabdomyolysis. Crit Care Med 1989;17:1073-4.
27. Flaque-Coma J. Cocaine and rhabdomyolysis: report of a case and review of the literature. Bol Asoc Med P R 1990;82:423-4.
28. Justiniani FR, Cabeza C, Miller BA. Cocaine-associated rhabdomyolysis and hemoptysis mimicking pulmonary embolism. Am J Med 1990;88:316-7.
29. Wholey M, Ogasawara E, Ramadan M. Acute rhabdomyolysis associated with cocaine intoxication: a case report. Hawaii Med J 1990;49:386-7.
30. Enriquez R, Palacios FO, Amoros FA, Cabezuelo JB, Hernandez F. Skin vasculitis, hypokalemia and acute renal failure in rhabdomyolysis associated with cocaine. Nephron 1991;59:336-7.
31. Horst E, Bennet RL, Barret O. Recurrent rhabdomyolysis in association with cocaine use. South Med J 1991;84:269-70.
32. Nolte KB. Rhabdomyolysis associated with cocaine abuse. Hum Pathol 1991;22:1141-5.
33. Adlakha A, Lobl J. A crack addict with extremely high creatine kinase levels [review]. Hosp Pract 1993;28:133-6.
34. Censori B, Camerlingo M, Casto L, et al. Acute rhabdomyolysis associated with acute cocaine intoxication: a case report. Ital J Neurol Sci 1993;14:325-7.
35. Daras M, Kakkouras L, Tuchman AJ, Koppel BS. Rhabdomyolysis and hyperthermia after cocaine abuse: a variant of the neuroleptic malignant syndrome? Acta Neurol Scand 1995;92:161-5.
36. Lampley E, Williams S, Myers S. Cocaine-associated rhabdomyolysis causing renal failure in pregnancy. Obstet Gynecol 1996;87:804-6.
37. Roth MD, Alarcon FJ, Fernandez JA, Preston RA, Bourgoignie JJ. Acute rhabdomyolysis associated with cocaine intoxication. N Engl J Med 1988;319:673-7.
38. Rubin R, Neugarten J. Cocaine-induced rhabdomyolysis masquerading as myocardial ischemia. Am J Med 1989;86:551-3.
39. Ahijado F, de Vinuesa S, Luno J. Acute renal failure and rhabdomyolysis following cocaine abuse. Nephron 1990;54:268.
40. Brody SL, Wrenn KD, Wilber MM, Slovis CM. Predicting the severity of cocaine-associated rhabdomyolysis. Ann Emerg Med 1990;19:1137-43.
41. Singhal PC, Rubin RB, Peters A, Santiago A, Neugarten J. Rhabdomyolysis and acute renal failure associated with cocaine abuse. Clin Toxicol 1990;28:321-30.
42. Welch R, Todd K, Krause G. Incidence of cocaine-associated rhabdomyolysis. Ann Emerg Med 1991;20:154-7.
43. SAS Institute. SAS procedures guide, version 6, 3rd ed. Cary, NC: SAS Institute, Inc., 1990.
44. U.S. Census Bureau. 1990 Census lookup (via Internet @http://venus.census.gov/cdrom/lookup.
45. Hamid A. The developmental cycle of a drug epidemic: the cocaine smoking epidemic of 1981-91. J Psychoactive Drugs 1992;24:337-48.
46. Wallace BC. Crack cocaine: a practical treatment approach for the chemically dependent. New York: Brunner/Mazel, 1991.
47. Inciardi JA. Editor's introduction: the crack epidemic revisited. J Psychoactive Drugs 1992;24:305-6.
48. Gugliotta G, Leen J. Kings of cocaine: inside the Medellin Cartel-an astonishing true story of murder, money, and international corruption. New York: Simon and Schuster, 1989.
49. Vanek V, Dickey-White H, Signs S, Schechter M, Buss T, Kulics A. Concurrent use of cocaine and alcohol by patients treated in the emergency department. Ann Emerg Med 1996;28:508-14.
50. Pagala M, Amaladevi B, Azad D, et al. Effect of cocaine on leakage of creatine kinase from isolated fast and slow muscles of rat. Life Sci 1993;52:751-6.
51. Brazeau G, McArdle A, Jackson M. Effects of cocaine on leakage of creatine kinase from skeletal muscle: in vitro and in vivo studies in mice. Life Sci 1995;57:1569-78.
52. Mann S, Boger W. Psychotropic drugs, summer heat and humidity, and hyperpyrexia: a danger restated. Am J Psychiatry 1978;135:1097-100.
53. Zelman S, Guillan R. Heat stroke in phenothiazine-treated patients: a report of three fatalities. Am J Psychiatry 1979;126:1787-90.
54. Keck PE, Pope HG, Cohen BM, McElroy SL, Nierenberg AA. Risk factors for neuroleptic malignant syndrome. Arch Gen Psychiatry 1989;46:914-8.
55. Farfel G, Kleven M, Woolverton W, Seiden L, Perry B. Effects of repeated injections of cocaine on catecholamine receptor binding sites, dopamine transporter binding sites and behavior in rhesus monkey. Brain Res 1992;578:235-43.
56. Cerruti C, Pilotte N, Uhl G, Kuhar M. Reduction in dopamine transporter mRNA after cessation of repeated cocaine administration. Mol Brain Res 1994;22:132-8.
57. Hitri A, Casanova M, Kleinman J, Wyatt R. Fewer dopamine transporter receptors in the prefrontal cortex of cocaine users. Am J Psychiatry 1994;151:1074-6.
58. Claye L, Akunne H, Davis M, DeMattos S, Soliman K. Behavioral and neurochemical changes in the dopaminergic system after repeated cocaine administration. Mol Neurobiol 1995;11:55-66.
59. Hitri A, Little K, Ellinwood E. Effect of cocaine on dopamine transporter receptors depends on routes of chronic cocaine administration. Neuropsychopharmacology 1996;14:205-10.
60. Tsukuda H, Kreuter J, Maggos C, et al. Effects of binge pattern cocaine administration on dopamine D1 and D2 receptors in the rat brain: an in vivo study using positron emission tomography. J Neurosci 1996;16:7670-7.
61. Seeman P, Van Tol H. Dopamine receptor pharmacology. Trends Pharmacol Sci 1994;15:264-70.
62. Wetli CV, Mash D, Karch SB. Cocaine-associated agitated delirium and the neuroleptic malignant syndrome. Am J Emerg Med 1996;14:425-8.
63. Staley J, Hearn W, Ruttenber A, Wetli C, Mash D. High affinity cocaine recognition sites on the dopamine transporter are elevated in fatal cocaine overdose victims. J Pharmacol Exp Ther 1994;271:1678-85.
64. Staley J, Wetli C, Ruttenber A, Hearn W, Mash D. Altered dopaminergic markers in cocaine psychosis and sudden death. NIDA Res Monogr 1994;153:491-504.
65. Warrian W, Halikas J, Crosby R, Carlson G, Crea F. Observations on increased CPK levels in "asymptomatic" cocaine abusers. J Addict Dis 1992;11:83-95.
66. Swartz CM, Breen KJ. Elevated serum CK in long abstinent cocaine abusers. Am J Drug Alcohol Abuse 1993;19:327-35.
Keywords:
Cocaine; Rhabdomyolysis; Excited delirium; Epidemiology

 Email Charly at: c-d-miller@neb.rr.com
Email Charly at: c-d-miller@neb.rr.com